Physics For Scientists And Engineers 6E - part 196
SECTION 25.7 • The Millikan Oil-Drop Experiment
781
these free electrons with the ionized air molecules. If a conductor has an irregular
shape, the electric field can be very high near sharp points or edges of the conductor;
consequently, the ionization process and corona discharge are most likely to occur
around such points.
Corona discharge is used in the electrical transmission industry to locate broken or
faulty components. For example, a broken insulator on a transmission tower has sharp
edges where corona discharge is likely to occur. Similarly, corona discharge will occur
at the sharp end of a broken conductor strand. Observation of these discharges is diffi-
cult because the visible radiation emitted is weak and most of the radiation is in the
ultraviolet. (We will discuss ultraviolet radiation and other portions of the electromag-
netic spectrum in Section 34.6.) Even use of traditional ultraviolet cameras is of little
help because the radiation from the corona discharge is overwhelmed by ultraviolet
radiation from the Sun. Newly developed dual-spectrum devices combine a narrow-
band ultraviolet camera with a visible light camera to show a daylight view of the
corona discharge in the actual location on the transmission tower or cable. The
ultraviolet part of the camera is designed to operate in a wavelength range in which
radiation from the Sun is very weak.
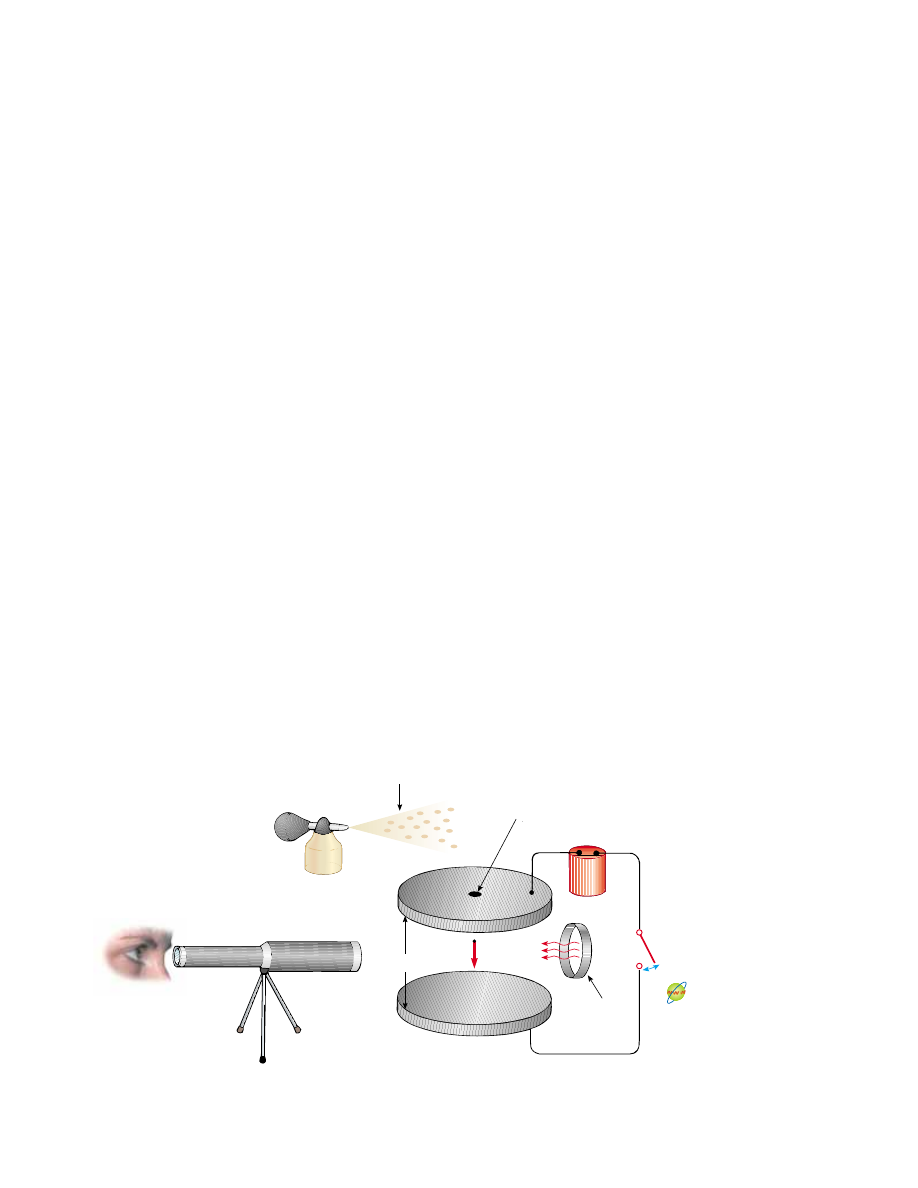
25.7 The Millikan Oil-Drop Experiment
During the period from 1909 to 1913, Robert Millikan performed a brilliant set of
experiments in which he measured e, the magnitude of the elementary charge on an
electron, and demonstrated the quantized nature of this charge. His apparatus,
diagrammed in Figure 25.27, contains two parallel metallic plates. Oil droplets from
an atomizer are allowed to pass through a small hole in the upper plate. Millikan used
x-rays to ionize the air in the chamber, so that freed electrons would adhere to the oil
drops, giving them a negative charge. A horizontally directed light beam is used to
illuminate the oil droplets, which are viewed through a telescope whose long axis is
perpendicular to the light beam. When the droplets are viewed in this manner, they
appear as shining stars against a dark background, and the rate at which individual
drops fall can be determined.
Let us assume that a single drop having a mass m and carrying a charge q is being
viewed and that its charge is negative. If no electric field is present between the plates,
Telescope
Oil droplets
Illumination
Pin hole
d
q
v
+
–
Active Figure 25.27 Schematic drawing of the Millikan oil-drop apparatus.
At the Active Figures link
at http://www.pse6.com, you
can do a simplified version of
the experiment for yourself. You
will be able to take data on a
number of oil drops and deter-
mine the elementary charge
from your data.